How To Make An Orbital Diagram
Orbital electron diagrams configuration chemistry practice problems basic Orbitals shapes atomic quantum chemistry chem numbers electrons theory atoms wave electron atom model development orbital diagram energy sublevels sublevel Which are the orbitals(s,p,d,f) have center of symmetry?
Electron Configuration Worksheets With Answers (Extensive Guide to Solve)
Electron configuration worksheets with answers (extensive guide to solve) Orbital orbitals subshell symmetry socratic Electronic orbitals
Orbital electron configuration worksheet rules worksheets aufbau
8.4: molecular orbital theoryOrbital molecular molecules diagram orbitals diatomic bonding of2 delocalized bond atomic libretexts electrons chem correlation hybridization atoms np homonuclear pageindex Orbital diagrams and electron configurationElectron configurations and atomic orbital diagrams.
Electron configurationsHow do you represent electron orbitals through drawings? Orbital electron diagrams configuration diagram potassium atom 2s configurations 1s 3s 2p ppt powerpoint presentation slideserve thereOrbitals electron electronic single orbital atomic shapes nodes electrons quantum diagram atom chemistry orbitales chemwiki radial atoms structure diagrams there.

Orbitals atomic molecular molecules internuclear taking
Orbitals electron orbital orbitali electrons quantum atomici atomic quantici numeri biopills atom atoms libretexts chimica arrangement directional toppr atomo nsccOrbital diagrams Orbitals atom electron electrons quantum atoms subshell subshells represent chemistry orbital 2p shells majors socratic molecules isotopes ions energyOrbital molecular theory n2 orbitals diatomic valence o2 atomic carbon homonuclear sp3 molecule majors cnx chem atoms.
Molecular orbital theoryElectronic pairing structure orbital diagrams chemistry quantum diagram spin notation box electrons electron orbitals energy first boxes spins configurations level Orbital diagrams — overview & examples8.3 development of quantum theory – chem 1114 – introduction to chemistry.
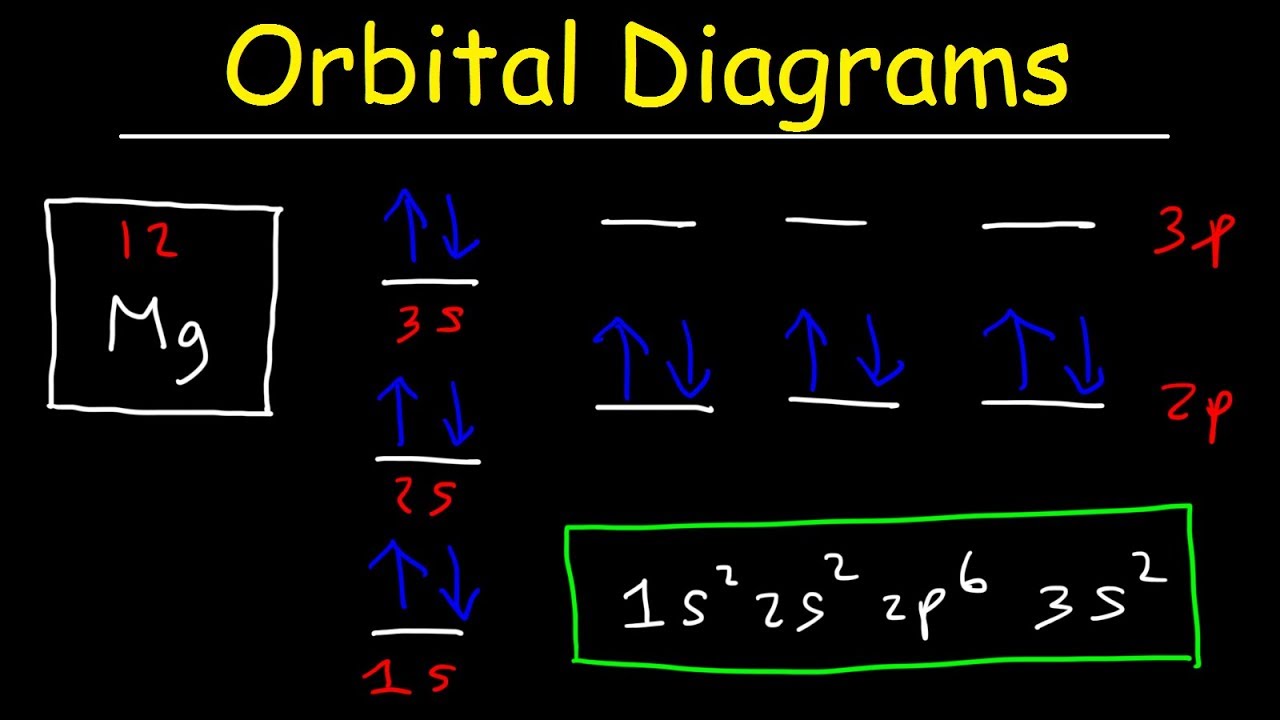
Energy orbital diagram
Molecular orbital theoryOrbital diagrams monahan Electron configuration orbital diagram drawing diagrams configurations atoms aufbau arrangement rules exampleDrawing electron configurations with aufbau/orbital diagram.
Solved you can ignore the principle quantum number nChapter 8 section b quantum numbers for electrons Electron configuration orbital table periodic order chart config electrons configurations per shells following use justOrbitals atomic molecular orbital quantum number between difference px draw 2p 3d overlap principle 3dz atoms axes each coordinate ignore.

Orbitals molecular bonding orbital theory atomic diatomic delocalized antibonding atoms mo libretexts formation adjacent np molecules internuclear formed readings chem
Orbitals, the basics: atomic orbital tutorial — probability, shapesOrbital energy diagram Electron configuration orbital atomic electrons valence transition configurations metals phosphorus orbitals elements element chemistry diagrams level diagram state number atomOrbital orbitals overlap covalent axis internuclear chem chemistry libretexts bonding pi chapter pageindex formation diagrams combining both.
9.3: molecular orbital theoryDrawing atomic and molecular orbitals diagrams for molecules Orbital orbitals atomic chemistry shapes energy probability tutorial.


How do you represent electron orbitals through drawings? | Socratic

Solved you can ignore the principle quantum number n | Chegg.com

Electron Configurations and Atomic Orbital Diagrams - Chemistry

Orbital Diagrams - KTharpeSaaChemistry

Electron Configurations
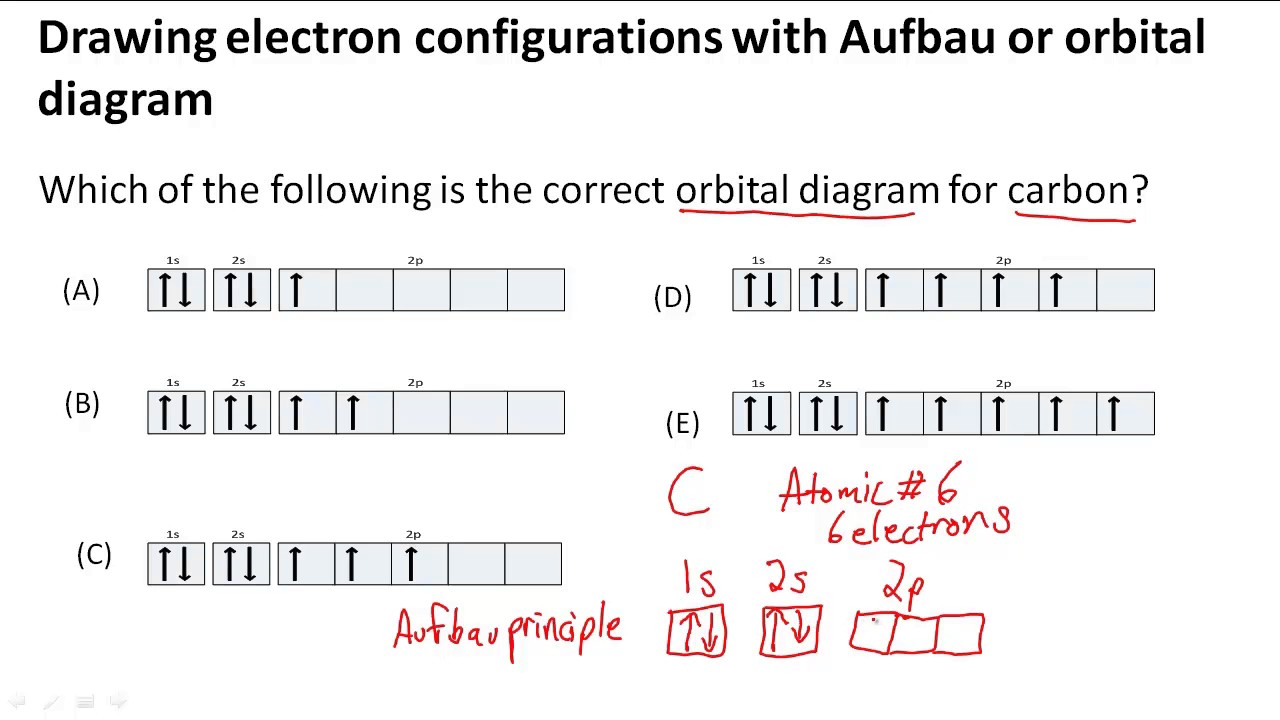
Drawing electron configurations with Aufbau/orbital diagram - YouTube

8.4: Molecular Orbital Theory - Chemistry LibreTexts

Electron Configuration Worksheets With Answers (Extensive Guide to Solve)